(This article was originally published on EurekAlert!, on 25 March 2026)
A research team led by Prof. SUN Jianwei, Chair Professor of the Department of Chemistry and Director of the Hong Kong Branch of Chinese National Engineering Research Centre for Tissue Restoration and Reconstruction at The Hong Kong University of Science and Technology (HKUST), has achieved a groundbreaking advance in organic synthesis and medicinal chemistry. The team has developed an air-stable chiral phosphine-catalyzed enantioselective approach to synthesize enantioenriched S(IV)-stereogenic vinyl sulfinamides—an under-explored class of organosulfur compounds with promising antiviral activity.
The importance of chiral-at-sulfur compounds in drug discovery and organic synthesis is indisputable. Over a quarter of top-selling small molecule pharmaceuticals contain sulfur atoms, and chiral sulfinamides bearing S(IV) chirality are key building blocks for medicinal chemistry, asymmetric synthesis auxiliaries, and catalytic ligands. However, current methods to access enantioenriched sulfinamides rely on transition metal catalysis with organometallic nucleophiles, and efficient organocatalytic strategies have long remained unexplored, creating a critical gap in synthetic chemistry for this valuable chemical space.
To address this challenge, Prof. Sun’s team published a landmark study in Nature Chemistry detailing the design and synthesis of a novel C₂-symmetric chiral phosphine catalyst—QianPhos—derived from the SPHENOL chiral skeleton. This custom catalyst exhibits extraordinary air stability and structural rigidity, which enables highly chemo-, enantio-, and diastereoselective C−S bond formation via a [3+2] annulation between Morita–Baylis–Hillman (MBH) esters and sulfinylamines. Unlike conventional transition metal-catalyzed approaches, this organocatalytic method generates in situ phosphorus ylides as vinyl nucleophiles, representing a mechanistically distinct route to chiral cyclic vinyl sulfinamides with excellent enantiopurity. Notably, these cyclic vinyl sulfinamides bind potently to the mutant SARS-CoV-2 spike protein and HIV-1 ENV protein, highlighting the great antiviral drug development potential of this under-explored chemical space.
Through a combination of density functional theory (DFT) calculations and mechanistic experiments (including ³¹P and ¹⁹F NMR spectroscopy), the team uncovered key mechanistic insights: the phosphonium species acts as the catalyst resting state, and sulfinylamines play a dual role as both reaction partners and promoters for the formation of the key catalytic intermediate—an unreported mechanistic feature that underpins the reaction’s high selectivity.
A group photo of the research team led by Prof. SUN Jianwei, Chair Professor of the Department of Chemistry and Director of the Hong Kong Branch of Chinese National Engineering Research Centre for Tissue Restoration and Reconstruction at HKUST (middle).
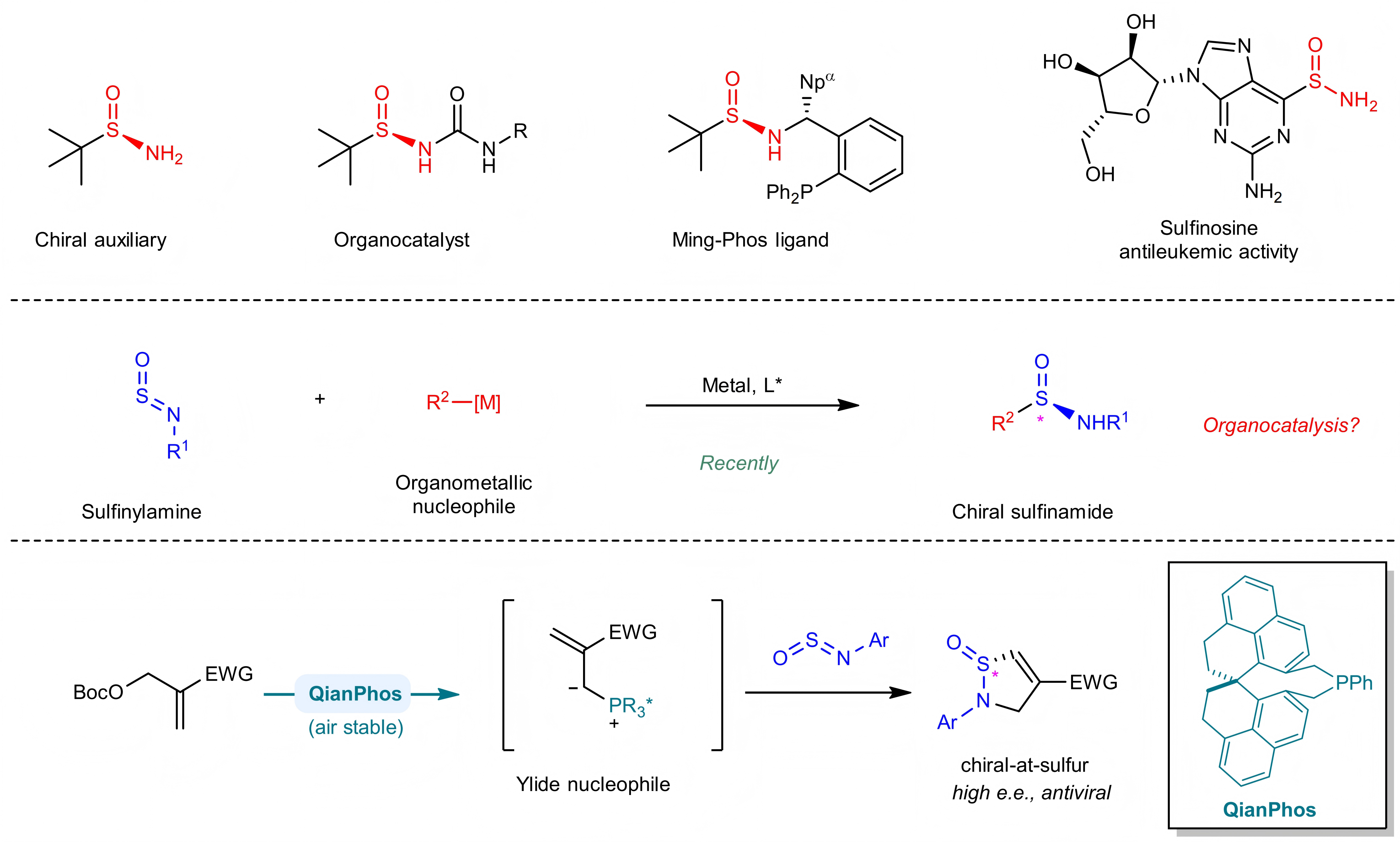
A graphic abstract of this study.



