(This article was originally published on EurekAlert! on May 10, 2024)
A research team led by the Hong Kong University of Science and Technology (HKUST) has discovered how carboxysomes, carbon-fixing structures found in some bacteria and algae, work. The breakthrough could help scientists redesign and repurpose the structures to enable plants to convert sunlight into more energy, paving the way for improved photosynthesis efficiency, potentially increasing the global food supply and mitigating global warming.
Carboxysomes are tiny compartments in certain bacteria and algae that encase particular enzymes in a shell made of proteins. They perform carbon fixation, which is the process of converting carbon dioxide from the atmosphere into organic compounds that can be used by the cell for growth and energy. Scientists have been trying to figure out how these compartments put themselves together.
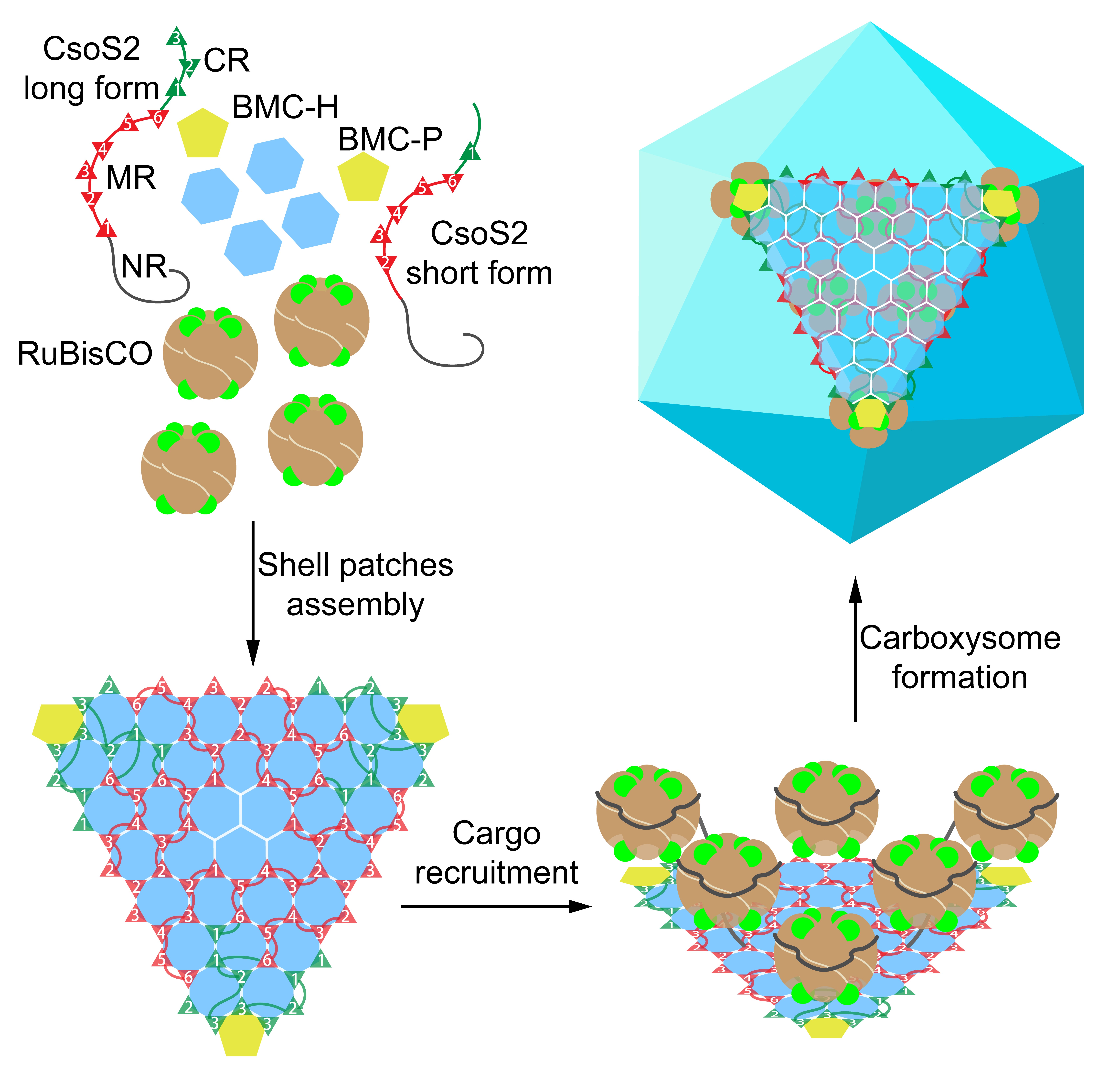
In their latest research, the team led by Prof. ZENG Qinglu, Associate Professor at HKUST’s Department of Ocean Science showed the overall architecture of carboxysomes purified from a type of bacteria called Prochlorococcus. In collaboration with Prof. ZHOU Cong-Zhao of the School of Life Sciences in the University of Science & Technology of China, the team overcame one of the biggest technical difficulties in cell breakage and contamination, which would prevent the proper purification of carboxysomes. The team also proposes a complete assembly model of α-carboxysome, which has not been observed in previous studies.
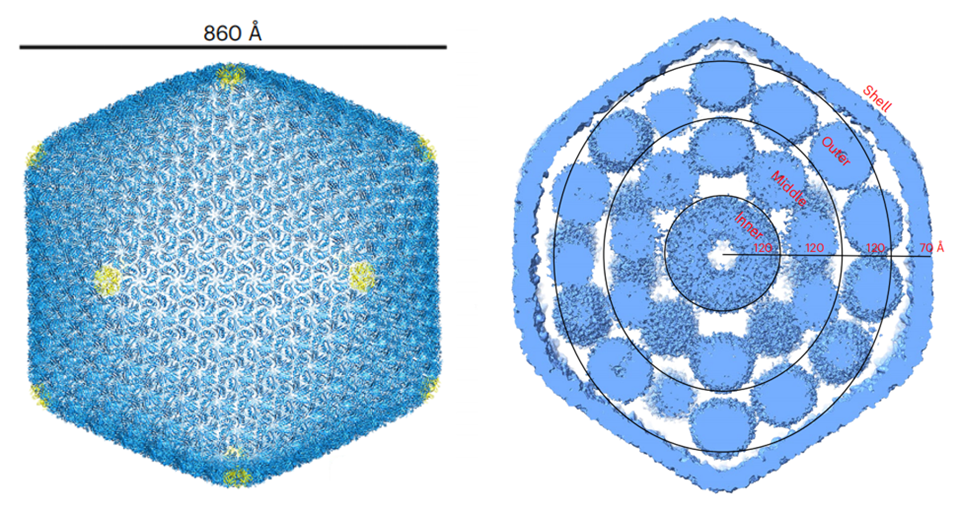
In specific, the team utilized the single-particle cryo-electron microscopy to determine the structure of α-carboxysome and characterize the assembly pattern of the protein shell, which looks like a 20-sided shape with specific proteins arranged on its surface. To obtain the structure of the minimal α-carboxysome with a diameter of 86 nm, they collected over 23,400 images taken from the microscope at the HKUST Biological Cryo-EM Center and manually picked about 32,000 intact α-carboxysome particles for analysis. Inside, the RuBisCO enzymes are arranged in three concentric layers, and the research team also discovered that a protein called CsoS2 helps to hold everything together inside the shell. Finally, the findings suggest that carboxysomes are put together from the outside in. This means that the inside surface of the shell is strengthened by certain parts of the CsoS2 protein, while other parts of the protein attract the RuBisCO enzymes and organize them into layers.
One of the most promising application of carboxysome is in plant synthetic biology, whereby the introduction of carboxysome into plant chloroplasts as the CO2-concentrating mechanism can improve photosynthetic efficiency and crop yield. “Our study unveils the mystery of α-carboxysome assembly from Prochlorococcus, thus providing novel insights into global carbon cycling,” says Prof. Zeng. The findings will also be important to slow down global warming, he says, as marine cyanobacteria fix 25% of global CO2. “Our understanding of the CO2 fixation mechanism of marine cyanobacteria will enable us to improve their CO2 fixation rate so that more CO2 can be removed from the atmosphere,” he says.
Following this study, the team plans to introduce Prochlorococcus α-carboxysome into plant chloroplasts and investigate whether the minimal α-carboxysome can improve the photosynthetic efficiency in plants. They also plan to modify the carboxysome genes and make genetically modified super cyanobacteria that are able to fix carbon dioxide at very high rates, which may be able to slow down global warming.
The study was recently published in the scientific journal Nature Plants.